The science is rarely the issue
Clinical trials don't slow down because teams lack rigor.
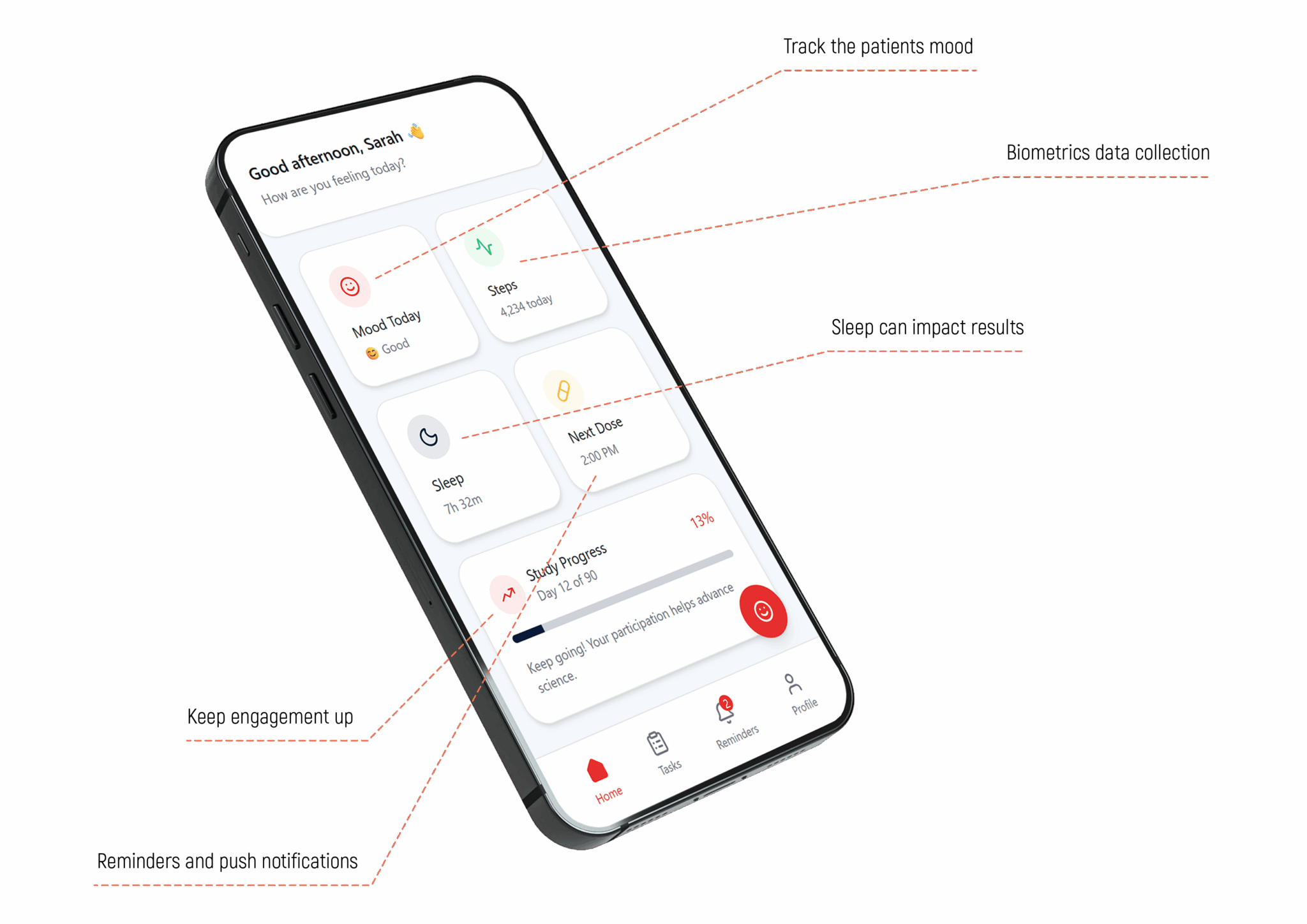
They slow down because participation is hard to sustain.
- Patients are asked to reshape their lives
- Sites chase incomplete or inconsistent data
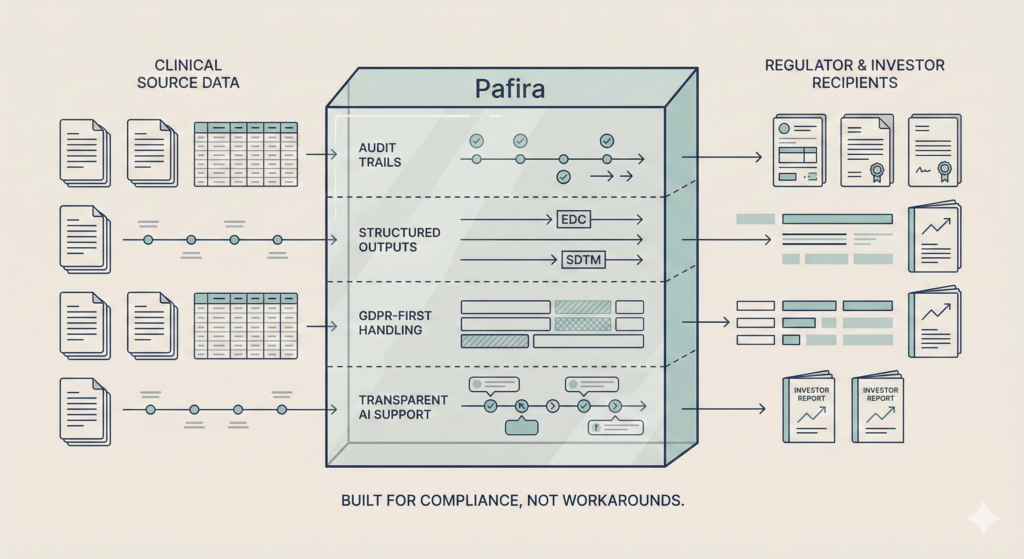
- Sponsors wait too long to see what's really happening
The result: delayed decisions, recruitment failure, and avoidable risk.